
This month is Head and Neck Squamous Cell Carcinoma (HNSCC) Awareness Month. There have been many developments in HNSCC, including a better understanding of its causes, as well as advances in efforts to apply genetics to help direct treatment.
The main treatment for HNSCC involves intensive radiation and chemotherapy. Among cancers treated with radiation, HNSCC is associated with some of the most challenging side effects. Reducing treatment-related toxicity is an area with a clear need for improvement. Unfortunately, HNSCC is often an aggressive disease with poor survival outcomes, making it equally important to consider the risk of cancer failure.
In this blog, we will share updates on human papilloma virus (HPV) infections as a cause of HNSCC, as well as MiraKind’s research on biomarkers to predict treatment outcomes and side effect risks for these patients.
What is head and neck cancer?
HNSCC is cancer that develops in the thin lining of the head and throat, including the mouth, back of the throat, and voice box. These are the most common cancers found in the head and neck region.
Areas affected by HNSCC
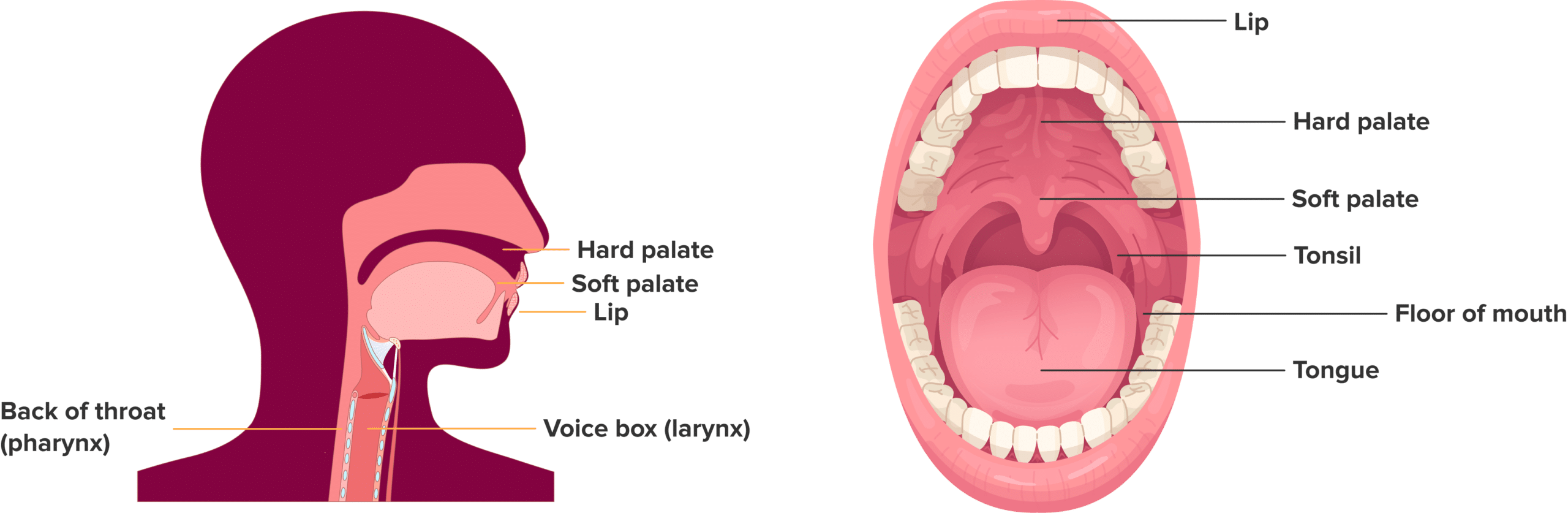
The highlighted areas show where HNSCC can develop. Adapted from Johnson DE et al., Head and neck squamous cell carcinoma, Nature Reviews Disease Primers 6, 92 (2020). Available at PubMed Central (PMCID: PMC7944998). Licensed under CC BY 4.0.
Changing risk factors and increasing cases for HNSCC
Risks for developing HNSCC have changed significantly over the past few decades. These cancers were once primarily caused by tobacco and alcohol use, but today about 30% of cases are caused by infection with certain strains of HPV.1
According to the American Cancer Society, the rate of HNSCC has been increasing steadily at about 0.7% per year over the past decade due in part to HPV cases.2
Patients with HPV-associated HNSCC tend to be younger, generally healthy, and often non-smokers. Changes in sexual behavior may be a contributor to this growing risk.3,4 In fact, HPV is the most common sexually transmitted infection in the United States, with about 13 million new cases each year.5 It is estimated that 80–90% of adults will be infected with HPV at some point in their lives, and most won’t even know it.6
Not all HPV infections lead to cancer. Certain high-risk types can disrupt the normal controls that keep cell growth in check.7 Over many years, this disruption can cause infected cells to become cancerous.8
Prognosis in Locally Advanced HNSCC
About 40% of patients are diagnosed with HNSCC at an early stage and can be cured with surgery or radiation alone.9 Unfortunately though, because early symptoms often resemble a cold or allergies, diagnosis is frequently delayed, and more than 60% of patients are found to have locally advanced HNSCC at the time of diagnosis.9
However, among these advanced cases, patients with HPV-positive cancers tend to respond better to the standard treatment of radiation plus chemotherapy, referred to as chemoradiation. In a study of HNSCC of all stages, the three-year survival of patients with HPV-positive HNSCC was approximately 80%, compared with only 50% for those with HPV-negative HNSCC.10
Standard treatment and side effects in HNSCC
In the late 1990s, some of the first clinical trials came out showing improved outcomes for HNSCC patients treated with a combination of chemotherapy, mainly cisplatin, and radiation as compared to radiation alone.11 This set the stage for chemoradiation to become the standard treatment for HNSCC.12
Among cancers treated with radiation, HNSCC has some of the most challenging side effects, especially when used in combination with chemotherapy. Chemotherapy can amplify the effects of radiation, such as controlling tumor growth and spread, but can also increase both acute and late toxicities.9,13,14
- Acute toxicity, which is experienced during or within the first 3–6 months after radiation, affects about 90% of HNSCC patients, with roughly 50% experiencing severe side effects, some of which can lead to treatment being interrupted. It includes mucositis, which causes painful mouth sores and ulcers that can make eating and drinking difficult, often requiring a feeding tube.15
- Late toxicity, which is experienced 6 months or later after radiation, affects about 50% of HNSCC patients. A common example is dysphagia, caused by scarring and stiffening of the muscles, tissues, and nerves that affect swallowing and speech, which can make daily life very challenging.16
Because side effects from treatment for HNSCC can be so difficult, there has been great interest in finding ways to identify patients that might be able to have less intensive treatment. This is called “de-escalation.”
The road to de-escalated treatments for HNSCC patients
In an effort to be able to de-escalate treatment, there has been a search for biomarkers, biological clues that can help identify which patients may respond just as well to less intensive treatment, allowing therapy to be safely de-escalated to minimize side effects. There has been an especially strong interest in finding de-escalation strategies for HPV-positive HNSCC patients, as they tend to be younger and seem to have better outcomes.
EGFR as a biomarker
Epidermal growth factor receptor (EGFR) is present in almost 90% of HNSCC cases.17 Cancers with high levels of EGFR tend to be more aggressive with a poorer response to treatment.18 Radiation therapy can sometimes increase the amount of this biomarker in the tumor, making it harder to treat.
Cetuximab, a drug that blocks the action of EGFR, was tested with radiation or chemotherapy. Early studies suggested it helped improve survival without adding significant side effects when compared to radiation or chemotherapy alone.19,20 This led researchers to try combining cetuximab with standard chemoradiation.
A large clinical trial, RTOG 0522, tested this approach. The results were disappointing: adding cetuximab to chemoradiation did not improve patient outcomes, regardless of EGFR levels.21 This suggests that EGFR alone is not a reliable biomarker and that standard chemoradiation remains the most effective treatment for HNSCC patients.
Today, for HNSCC, the main treatment is chemoradiation, while cetuximab is used in combination with radiation only if the patient is unable to tolerate chemotherapy.
But what about HPV-positive HNSCC patients? Is it possible that HPV status could be used as a potential biomarker to identify HNSCC patients where de-escalation is possible?
HPV as a biomarker
The potential of HPV to act as a biomarker allowing de-escalation was tested in two major trials—RTOG 1016 and De-ESCALaTE HPV. These trials were launched to test whether chemotherapy with radiation versus cetuximab with radiation (considered a less toxic combination) could safely reduce side effects with the same survival rate for HPV-positive HNSCC patients.
However, disappointingly, both trials found that cetuximab plus radiation was less effective than the standard of care, cisplatin plus radiation. Patients receiving radiation plus cetuximab had worse overall survival, higher rates of cancer recurrence, and no meaningful reduction in side effects compared to chemoradiation.22,23
What these findings suggest is that de-escalation for HPV-positive patients using cetuximab instead of cisplatin is not a safe approach. Although alternative therapies do exist,24 chemoradiation seems to be the most effective treatment for HPV-positive HNSCC as well.
The search for other reliable biomarkers
Unfortunately, even after years of research, treatment for HNSCC remains unchanged, and no biomarker can reliably predict outcomes or severe side effects to enable treatment personalization. As a result, patients must still undergo aggressive therapy, despite the risk of long-term complications.
If these biomarkers aren’t enough to predict who will respond to treatment or have more severe side effects, what could?
The answer to this may lie in your genetics.
MicroRNAs and cancer biology
MicroRNAs, or miRNAs, are small molecules that help regulate genetic information by controlling how much protein is made. You can think of them like a dial, turning protein production up or down so cells can handle stress, including damage from cancer treatments.
Some people inherit tiny changes in these molecules or in the spots where they attach, called mirSNPs. Even the tiniest change can influence how cells handle stress. This makes them a unique genetic biomarker that can help predict both treatment outcomes and potential side effects before therapy begins.
Proper vs. Reduced miRNA Binding
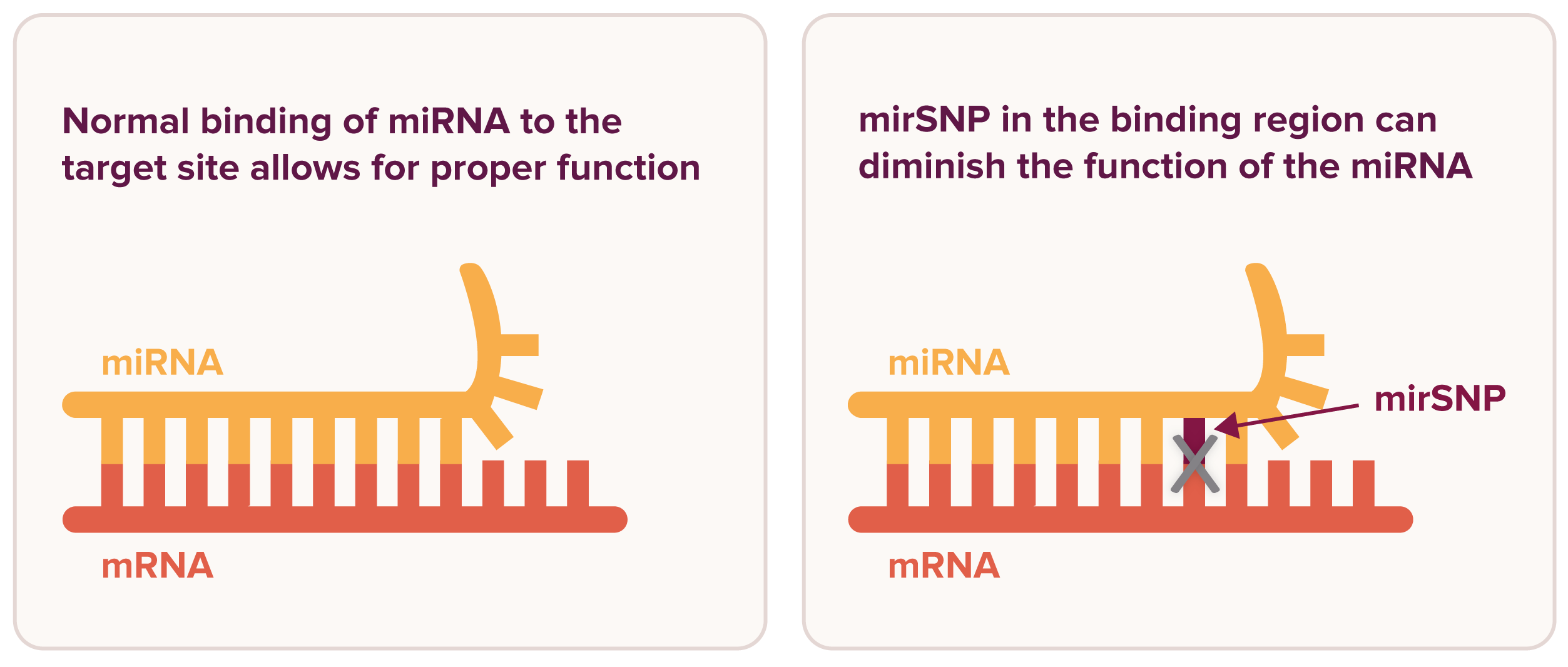
miRNAs influence gene expression by binding to messenger RNA (mRNA). A mirSNP in the miRNA binding site on the mRNA or in the miRNA itself can influence the body’s response to stress and cellular damage. Figure adapted from Malhotra et al., “Breast Cancer and miR-SNPs: The Importance of miR Germ-Line Genetics,” Non-Coding RNA, 2019, DOI: https://doi.org/10.3390/ncrna5010027. CC-BY 4.0 license.
KRAS-variant as a biomarker
The first example of a mirSNP in cancer was the KRAS-variant. Several studies have shown that patients with this variant often don’t respond well to chemotherapy but can do better with cetuximab.25-30
In a reanalysis of patients from the RTOG 0522 trial by Dr. Joanne Weidhaas, HNSCC patients with the KRAS-variant were shown to have better outcomes without worsened toxicity when cetuximab was added to chemoradiation. In contrast, patients without the KRAS-variant did not have improved outcomes and had worse toxicity with the addition of cetuximab.31
KRAS-variant status and treatment response

In this table, “chemoradiation” refers to the standard treatment, which combines cisplatin with radiation. This standard treatment is used as a baseline to compare how adding cetuximab affects outcomes and side effects.
These studies suggested there might be a meaningful link between the KRAS-variant and treatment response for HNSCC patients receiving cetuximab without worsening toxicity.
KRAS-variant and treatment outcomes and toxicity for HPV+ patients
This prompted a follow-up study to evaluate the KRAS-variants’ potential as a biomarker for predicting outcomes and toxicities in HPV-positive patients receiving radiation plus cetuximab versus radiation plus cisplatin in the RTOG 1016 trial.
In a reanalysis of samples from the trial, Dr. Weidhaas and her colleagues at UCLA found that, although the KRAS-variant did not predict better survival or lower recurrence, it appeared to influence treatment toxicity, or side effects.
In the original RTOG 1016 trial, HPV-positive patients receiving radiation plus cetuximab showed worse tumor control with no significant difference in overall toxicity. But when grouped based on KRAS-variant status, a clearer picture emerged: those with the variant did not experience worse tumor control, whereas those without the variant did. Additionally, KRAS-variant patients showed lower rates of acute toxicity but higher rates of severe late toxicity, although not statistically significant.32
KRAS-variant and treatment toxicity in RTOG 1016 trial

In this table, cisplatin + radiation refers to the standard treatment. This standard treatment is used as a baseline to compare how adding cetuximab affects outcomes and side effects.
Can a single mirSNP, such as the KRAS-variant, predict treatment-specific toxicity? The findings suggest it might, but its predictive power may be limited on its own.
Strength in numbers: mirSNP signatures
Combining several mirSNPs into a single signature improves their ability to predict treatment side effects and outcomes. For example, the team’s earlier work with PROSTOX showed that two distinct signatures could predict the risk of late toxicity, each corresponding to a different radiation regimen in prostate cancer.33,34 In a follow-up study, they identified a different signature that predicted acute toxicity.35
Building on these findings, the team now has applied a similar approach to HNSCC patients by further analyzing the RTOG 0522 study. Early results are encouraging: using mirSNP signatures seems to improve predictions of both short-term and long-term side effects from chemoradiation, as well as overall survival. The team will share these findings in the fall after reporting them at the annual ASTRO 2026 meeting in Boston!
Looking ahead: Personalized cancer care
The road to effective and less toxic treatments for locally advanced HNSCC has been long, but the outlook for patients with this cancer is becoming more promising. Research into mirSNP signatures has the potential to one day help clinicians identify patients who may be at higher risk of severe side effects, as well as those for whom de-escalation strategies are possible.
This personalized approach has the power to give patients and their care teams more information before treatment starts, which allows for treatment plans that are safer and more equally effective for each individual.
References
- https://www.mdpi.com/1999-4915/17/5/662
- https://www.cancer.org/content/dam/cancer-org/research/cancer-facts-and-statistics/annual-cancer-facts-and-figures/2025/2025-cancer-facts-and-figures-acs.pdf
- https://pmc.ncbi.nlm.nih.gov/articles/PMC8035131/
- https://pmc.ncbi.nlm.nih.gov/articles/PMC5012241/
- https://www.cdc.gov/hpv/about/index.html
- https://www.cdc.gov/sti/about/about-genital-hpv-infection.html
- https://www.sciencedirect.com/science/article/pii/S2666679021000033
- https://www.cell.com/cell-host-microbe/fulltext/S1931-3128(14)00069-9
- https://pmc.ncbi.nlm.nih.gov/articles/PMC10546034/
- https://pmc.ncbi.nlm.nih.gov/articles/PMC8497986/
- https://pubmed.ncbi.nlm.nih.gov/9619357/
- https://www.nature.com/articles/ncponc0750
- https://pubmed.ncbi.nlm.nih.gov/12506176/
- https://pmc.ncbi.nlm.nih.gov/articles/PMC4911537/
- https://pmc.ncbi.nlm.nih.gov/articles/PMC12666529/
- https://pmc.ncbi.nlm.nih.gov/articles/PMC7136308/
- https://pubmed.ncbi.nlm.nih.gov/16763281/
- https://pmc.ncbi.nlm.nih.gov/articles/PMC25480/
- https://www.nejm.org/doi/10.1056/NEJMoa053422
- https://www.nejm.org/doi/full/10.1056/NEJMoa0802656
- https://pmc.ncbi.nlm.nih.gov/articles/PMC4162493/
- https://pmc.ncbi.nlm.nih.gov/articles/PMC6541928/
- https://www.thelancet.com/journals/lancet/article/PIIS0140-6736%2818%2932752-1/fulltext
- https://ascopubs.org/doi/10.1200/EDBK_280687
- https://pmc.ncbi.nlm.nih.gov/articles/PMC3342446/
- https://pmc.ncbi.nlm.nih.gov/articles/PMC2691138/
- https://pmc.ncbi.nlm.nih.gov/articles/PMC4207729/
- https://www.sciencedirect.com/science/article/abs/pii/S1877782114001349?via%3Dihub
- https://pmc.ncbi.nlm.nih.gov/articles/PMC4045863/
- https://pmc.ncbi.nlm.nih.gov/articles/PMC4155520/
- https://jamanetwork.com/journals/jamaoncology/fullarticle/2594275
- https://pubmed.ncbi.nlm.nih.gov/41790036/
- https://pubmed.ncbi.nlm.nih.gov/34990726/
- https://pubmed.ncbi.nlm.nih.gov/36880064/
- https://aacrjournals.org/clincancerres/article/31/12/2530/762786/Validation-and-Derivation-of-miRNA-Based-Germline



Leave a Reply